Science/Health
Aspergillus ODO Recall Arizona: Full Report, Health Risks, and Updates
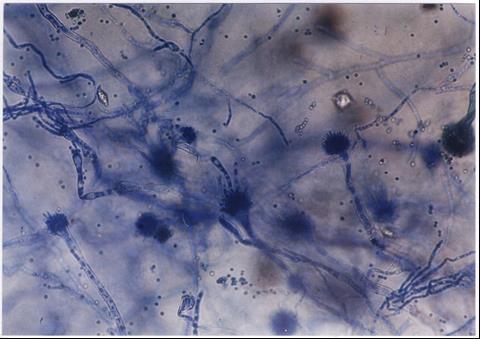
When the news broke about the aspergillus odo recall arizona, it caught the attention of both consumers and health officials across the state. What began as a routine quality check has now turned into a major public health alert. The recall revolves around a contaminated batch of products linked to Aspergillus fungus, a potentially harmful microorganism that can cause severe infections in vulnerable individuals. As Arizona’s Department of Health Services and the FDA step in, people are asking what this contamination means, who’s at risk, and how far the issue might spread.
The recall isn’t just a local problem. It’s a reminder of how one small lapse in testing or quality control can expose thousands to risk. Aspergillus contamination has triggered multiple recalls before, but the ODO case in Arizona in 2025 has raised bigger questions about supply chain oversight, product safety standards, and public health monitoring.
What Happened in the Aspergillus ODO Recall
The incident began when the aspergillus odo recall arizona identified contamination in a product line under the brand name ODO. Testing showed the possible presence of Aspergillus fungus in a batch coded PHX1164-ODO. Once results were confirmed, the company initiated a voluntary recall to prevent potential exposure. Health authorities made the announcement, advising all retailers and consumers to stop using and dispose of the affected items immediately.
The recall was categorized as a Class II recall, which means the contamination posed a moderate health risk but was not immediately life-threatening. Even so, any product that tests positive for Aspergillus contamination must be pulled to protect consumers, particularly those with weak immune systems or respiratory illnesses.
Understanding Aspergillus Contamination
Aspergillus is a common mold found in the environment, but when it contaminates consumer goods, it poses health risks that can range from mild allergic reactions to severe lung infections. The fungus spreads through microscopic spores that thrive in warm and humid environments. In normal conditions, most people breathe in these spores without harm. However, when large numbers of spores enter the lungs or when products are consumed, the risks multiply.
In Arizona’s dry climate, fungal contamination might seem unlikely, yet indoor cultivation facilities, warehouses, and packaging sites can easily become breeding grounds. If even one area becomes contaminated, spores can spread quickly through air systems, equipment, and raw materials. Once a contaminated product reaches consumers, inhalation or contact can trigger infection.
The Science Behind Aspergillus ODO Contamination
The species of Aspergillus most often associated with recalls are Aspergillus fumigatus, Aspergillus flavus, Aspergillus niger, and Aspergillus terreus. Each of these can cause respiratory infections known as aspergillosis. The fungus reproduces by releasing spores into the air, and these spores settle on surfaces or materials like food, herbs, or plant-based products.
During laboratory testing, contamination is identified through culture-based tests or molecular detection methods. In this case, the contaminated ODO batch failed microbial screening, indicating fungal presence. The contamination likely occurred during processing or storage, possibly due to improper humidity control, poor ventilation, or unsanitized equipment.
Why the Recall Was Issued
Recalls of this kind are meant to protect consumers before illnesses occur. The aspergillus odo recall arizona was triggered after lab tests detected fungal contamination above acceptable levels. While no confirmed illnesses have been linked to the ODO products so far, authorities took immediate precautionary action. When even a single batch fails microbial testing, every related lot must be reviewed and possibly recalled.
The Arizona Department of Health Services instructed dispensaries and retailers to remove all ODO products associated with the contaminated lot. Consumers who purchased the affected batches were told to stop using them and dispose of them safely. The recall notice emphasized that the action was voluntary but necessary to maintain consumer safety and product integrity.
Health Risks Linked to Aspergillus ODO Contamination
The fungus at the center of the recall poses different levels of threat depending on individual health conditions. People with healthy immune systems might only experience mild allergic reactions. But for those with asthma, chronic lung conditions, or weakened immunity, exposure could lead to serious complications.
Common symptoms include coughing, wheezing, chest pain, shortness of breath, and fatigue. In more severe cases, invasive aspergillosis can occur, where the fungus spreads from the lungs to other parts of the body like the brain, heart, or kidneys. If not treated, it can be fatal.
Because of these risks, the recall stressed the need for awareness and immediate action. Even if no illness reports have emerged, the danger lies in potential delayed infections, where symptoms may appear weeks after exposure.
FDA Involvement and Oversight
The U.S. Food and Drug Administration became involved in overseeing the recall and verifying that proper steps were being taken. FDA recall classifications help determine the severity and required response. In the case of the Aspergillus ODO contamination, the agency’s focus is on tracking affected products, ensuring removal from retail shelves, and verifying that corrective actions are in place to prevent recurrence.

FDA inspectors may conduct follow-up visits to facilities associated with the contaminated batch. Their inspection process typically reviews microbial testing records, environmental monitoring logs, and sanitation procedures. If violations are found, the manufacturer could face penalties or further enforcement actions.
Arizona’s Public Health Response
The Arizona Department of Health Services (ADHS) moved quickly once the contamination was detected. The agency’s Consumer Safety Division published a public notice to alert buyers and issued a product disposal guide. Retailers were also required to report how many units of the ODO batch had been sold and whether they had been removed from inventory.
ADHS teams have since been investigating potential contamination sources across storage facilities and processing plants. Early findings suggest possible humidity issues or ventilation failures during product drying or packaging. Officials have also expanded testing to nearby facilities to rule out cross-contamination.
Possible Sources of Aspergillus ODO Contamination
While the final contamination report hasn’t been fully disclosed, experts believe several points in the supply chain could be responsible.
- Cultivation Environments: If the raw plant material was grown in a humid or unsterile environment, fungal spores could have developed naturally.
- Processing Equipment: Contaminated machinery or drying areas might have spread spores onto clean products.
- Storage Conditions: Warm, damp warehouses can allow Aspergillus spores to grow rapidly, especially if airflow is poor.
- Packaging and Handling: Once sealed, a contaminated product can preserve spores, allowing them to survive and multiply until consumption.
Each of these factors is under review as part of the ongoing investigation.
The Timeline of the Aspergillus ODO Recall Arizona 2025
The sequence of events leading to the recall unfolded quickly.
- Early May 2025: Routine testing flagged potential microbial contamination in an ODO-branded batch.
- Mid-May 2025: Confirmatory lab tests detected Aspergillus spores.
- Late May 2025: ODO voluntarily recalled affected products, working with ADHS.
- June 2025: Public notice released urging consumers to discard products and monitor health.
- July 2025: FDA updates classified it as a contamination recall with no reported illnesses.
- November 2025: Investigations continue as ADHS and FDA review supplier compliance and possible cross-contamination risks.
This timeline shows how fast regulators can act once contamination is detected, but it also highlights how contamination can pass initial quality checks before being caught.
How Testing Works for Aspergillus
Testing for Aspergillus contamination involves multiple layers of screening. First, samples are cultured in lab environments that promote fungal growth. If colonies form, labs identify the species using DNA analysis or microscopic evaluation. Some testing methods use qPCR (quantitative polymerase chain reaction) to detect fungal DNA quickly and accurately.
Regulations in Arizona require all consumable plant-based products to undergo microbial testing before sale. However, lapses can happen. Equipment malfunction, human error, or contamination during sampling can all cause false results. That’s why follow-up testing and independent lab verification are critical.
Lessons from the Aspergillus ODO Recall
The 2025 Aspergillus ODO recall in Arizona underscores several important lessons. Product safety isn’t guaranteed just because a product passes initial tests. Quality control must be ongoing, and storage conditions must be closely monitored. Manufacturers must also understand that fungal contamination isn’t always visible—it can exist without any noticeable mold growth or odor.
The recall also shows the importance of transparency. Public awareness is vital in preventing exposure, yet many consumers only learn about recalls after days or weeks. Faster communication through online alerts, text notifications, and retailer outreach could reduce that delay.
Who’s Most at Risk
Certain groups face greater danger from Aspergillus exposure. Individuals with asthma, cystic fibrosis, chronic obstructive pulmonary disease (COPD), or immune-suppressive treatments (like chemotherapy or organ transplants) are more likely to develop severe infections. Pregnant women, elderly individuals, and people on long-term steroids are also more vulnerable.

For these groups, even limited exposure to contaminated products could trigger a medical emergency. Healthcare providers have been advised to remain alert for respiratory infections that may mimic other illnesses but could stem from Aspergillus contamination.
Consumer Steps After the Recall
If you purchased an ODO product in Arizona, the first step is to check batch numbers printed on packaging or receipts. Any batch linked to the recall—especially PHX1164-ODO—should be discarded immediately. Consumers should avoid using any remaining product even if it looks normal. The contamination may not be visible or detectable by smell.
Keep proof of purchase in case refund programs are offered by the manufacturer or retailer. If you’ve experienced respiratory symptoms after using these products, contact your doctor and mention potential Aspergillus exposure. It helps clinicians order the right diagnostic tests, such as chest scans or fungal cultures.
Industry Impact and Regulatory Changes
The Aspergillus ODO recall is already pushing Arizona’s product safety regulations toward stricter enforcement. ADHS is reportedly reviewing new rules that would require more frequent microbial testing and third-party audits for licensed manufacturers. Some industry observers predict that testing labs will need to revalidate their equipment and adopt new methods to detect contamination more reliably.
The recall has also spurred national discussions about fungal safety in inhalable products. Similar recalls in states like California, Michigan, and Colorado have shown that microbial contamination isn’t limited by geography or product category. As awareness grows, manufacturers are expected to adopt cleaner production practices, improved ventilation systems, and regular sanitation audits.
The Role of the FDA in Preventing Future Outbreaks
The FDA’s role extends beyond enforcement—it sets testing standards and recall procedures. After incidents like this, the agency usually issues updated guidance to the industry. This may include revised acceptable microbial limits, standardized testing timelines, and stronger penalties for non-compliance.
By coordinating with state agencies like ADHS, the FDA ensures consistent national responses to contamination incidents. In the case of the aspergillus odo recall arizona, their oversight ensures the recall is fully executed and that affected products are not redistributed under different packaging.
How Consumers Can Stay Updated
Consumers can stay informed through the FDA’s recall database, state health department announcements, and local news updates. Signing up for product safety alerts is also useful. Retailers and dispensaries in Arizona are required to post recall notices visibly at point-of-sale areas. Checking those notices regularly helps consumers avoid purchasing affected items.
Long-Term Implications for Public Health
The Aspergillus ODO contamination might appear as a localized issue, but its impact stretches far beyond Arizona. It highlights the fragile balance between industrial production and microbial safety. As more people turn to plant-based consumables and supplements, contamination monitoring will need to evolve. Recalls like this one will likely become lessons that shape future safety standards.
Public health agencies may also expand fungal surveillance programs to track exposure patterns and potential outbreaks. Fungal infections often go underreported, so this incident could motivate improved data collection and medical awareness.
Possible Future Developments
Experts predict that by early 2026, we’ll see comprehensive reports detailing the root cause of the Aspergillus ODO contamination. That report will likely include environmental assessments, employee safety audits, and equipment testing data. Depending on the findings, penalties or regulatory actions might follow.
Manufacturers connected to ODO could also be required to implement contamination prevention plans, including sterilization protocols, air filtration upgrades, and enhanced training for production staff. Future recalls might even be accompanied by real-time public dashboards showing contamination sources, product testing outcomes, and batch traceability.
Final Thoughts
The aspergillus odo recall arizona in Arizona isn’t just another contamination event—it’s a warning about how fragile product safety systems can be. When microbial risks slip through the cracks, consumers pay the price. Arizona’s quick response helped prevent potential illness, but the situation reveals deeper weaknesses in testing, transparency, and oversight.
For now, the best course of action for consumers is simple: check your products, stay informed, and take any recall notice seriously. Fungal contamination may sound minor, but when it involves Aspergillus, the risks can’t be ignored. Manufacturers, regulators, and consumers all share responsibility in ensuring contaminated goods never reach the shelves again.
Science/Health
SynapseX: Engineering the Future of Brain Health Intelligence

Brain health is not only about hospitals or old age. It shapes how we learn how we work and how we feel each day. When the brain is well supported people think clearly handle stress better and recover faster after hard days. When it is not supported even simple tasks can feel heavy.
Today we have better tools than ever before. Phones wearables and safe digital tests can help people track signals connected to sleep attention and recovery. When these signals are used responsibly they form brain health intelligence. That means using measured patterns to guide better daily choices and better conversations with professionals.
If you want to improve your focus and mental clarity with simple daily routines, read our in-depth guide, Train Your Brain for Peak Performance, Habits That Boost Focus and Mental Clarity.
SynapseX is a forward looking concept that aims to engineer this future with care. It focuses on clarity safety and trust so people can use insights without fear. This article explains what brain health intelligence is how it can help in real life and what responsible engineering should look like.
Why brain health matters now
Brain health is part of everyday life. It affects memory mood learning speed patience and motivation. It also affects how we communicate with family friends classmates and coworkers. In the past many people only thought about brain health after a big problem. Now more people want to prevent problems early and support good habits before things get worse.
Modern life can be intense. Many people sleep less than they need. Many feel pressure to respond quickly all day long. Screens are everywhere and quiet time is rare. Over time these pressures can reduce focus and make stress feel stronger.
Brain health intelligence can help because it helps people notice patterns early. It can show when sleep is slipping when recovery is weak and when workload is too high. That early view can help people act sooner.
Brain health affects school work and family life
Brain health shows up in simple moments.

- A student reads the same page many times and still cannot remember it
- An office worker feels tired even after eight hours in bed
- A parent gets irritated quickly because stress is high
- An older adult becomes less confident with routine tasks
These moments do not always mean a serious disorder. Often they are signs that sleep stress and routines need support. Brain health intelligence helps people see which habits may be causing the problem.
Small changes add up over time
Brain health usually changes slowly. That is good news because small improvements can also add up. A little more sleep consistency more movement and better breaks during work can improve energy and focus.
It is also why trends matter more than a single bad day. Everyone has off days. The goal is to understand direction. Are things improving staying stable or slowly getting worse. Brain health intelligence is useful when it helps people see that direction.
Understanding brain health intelligence
Brain health intelligence is the use of measured signals to understand brain related wellbeing over time. It does not replace doctors. It does not label people. It supports better choices by turning daily patterns into clear guidance.
Think of it like a simple dashboard. The dashboard does not control the car. It helps the driver understand what is happening so they can make better decisions.
Brain health intelligence becomes powerful when it is used consistently and responsibly. It can help people connect the dots between sleep stress workload and mental energy.
What brain health intelligence measures
Different systems measure different things. Common signals include:
- Sleep duration and sleep timing
- Sleep consistency across weekdays and weekends
- Daily activity and movement
- Short cognitive tasks such as reaction time and attention checks
- Optional self reports for mood stress and energy
- Some heart related signals such as heart rate variability as a stress linked measure
Each signal has limits. Wearables estimate sleep rather than measure it perfectly. Self reports depend on honesty and memory. Cognitive tasks can be affected by noise distractions and motivation. This is why a good system uses multiple signals and focuses on trends.
How insights become guidance
Data becomes useful when it leads to simple steps. Examples of helpful guidance include:
- Your sleep timing changed a lot this week consider a steady bedtime window
- Focus dips appear after late nights schedule hard tasks earlier tomorrow
- Stress is rising and recovery is falling consider a lighter day and a short walk
- Your trend looks different than usual consider checking in with a clinician if this continues
The goal is to support action without panic. A good system communicates uncertainty and avoids dramatic statements.
SynapseX and the next generation of brain insight
SynapseX aims to bring engineering discipline and user friendly design to brain support. The core idea is simple. Use responsible measurement to help people understand their brain health trends and guide better choices.

For SynapseX to be useful it must be easy to use and easy to trust. People should not need a medical degree to understand their own results. They should also feel safe about their data.
In a crowded market trust will be the deciding factor. People will choose tools that respect privacy avoid fear and show clear value.
Human first design
Human first design means the product fits real life. It should work for people who are busy who are not technical and who may be stressed already.
Human first design includes:
- Clear language at a Grade 7 reading level
- Short explanations for each metric
- A simple view of weekly trends
- Practical tips that are easy to try
- Accessibility options such as readable text and calm colors
It also includes cultural sensitivity. People have different routines and family duties. A one size approach often fails.
Supportive alerts and safe nudges
Alerts can help but they can also annoy or scare people. SynapseX should use nudges that feel calm and respectful.
Supportive nudges can include:
- A reminder to keep bedtime steady when sleep timing drifts
- A suggestion to take a short break after long screen time
- A prompt to do a one minute breathing exercise during a high stress day
- A suggestion to talk to a professional if the trend stays concerning
SynapseX should avoid acting like an alarm system. It should act like a steady coach.
The engineering behind trusted brain systems
Building brain tools requires more than a nice app design. It requires careful engineering. The system must handle data quality protect privacy and avoid false claims. It should also be tested in real conditions not only in lab demos.
Responsible engineering includes three big areas.
- Data collection and quality
- Modeling and interpretation
- Security privacy and governance
Passive and active data collection
Passive data is collected with little effort. It can include sleep estimates steps and device usage patterns if users allow it. Passive data is convenient but it can miss context.
Active data requires a user action. It can include short tasks short check ins and guided exercises. Active data gives more meaning but it requires motivation.
A balanced brain health intelligence system combines both.
Passive strengths
- Low effort
- Good for long term trends
- Works in the background
Passive limits
- Can be inaccurate on some nights
- Can confuse travel illness or new routines
- May not explain why a change happened
Active strengths
- Adds context and clarity
- Can measure focus directly with a simple task
- Helps users reflect and notice feelings
Active limits
- Users may skip when busy
- Can be influenced by distractions
AI basics with real limits

AI can find patterns across many signals. It can learn that certain combinations often appear before fatigue or attention drops. That helps the system offer better timing for suggestions.
Still AI is not magic. It does not know your whole life. It cannot see personal context unless you provide it. It also learns from data and data can be biased.
Good engineering practices include:
- Testing models across age groups and lifestyles
- Monitoring performance over time
- Avoiding medical diagnosis claims
- Showing confidence levels and uncertainty
- Allowing users to correct context such as travel illness exams or night shifts
The system should stay humble. When it is unsure it should say so.
Security and privacy by design
Brain related data is sensitive. People deserve strong protections. SynapseX should follow privacy by design principles from the start.
Key protections include:
- Consent that is clear and specific
- Data minimization collect only what is needed
- Encryption in transit and at rest
- Strong access controls and audit logs
- Clear retention rules and deletion options
- Safe sharing tools so users control who sees their data
Trust grows when users feel in control.
Real life use cases
Brain health intelligence matters when it helps people in daily life. Below are practical use cases where SynapseX could provide value in a safe responsible way.
Sleep recovery and energy
Sleep is one of the strongest drivers of brain performance. Many people focus only on hours. But consistency also matters. Going to bed at random times can lead to tired days even if total hours look fine.
SynapseX could support sleep by:
- Showing sleep timing consistency
- Highlighting recovery trends across the week
- Offering small routine suggestions
- Helping users plan for late nights by improving recovery the next day
Practical tips that a system can reinforce include:
- Keep a steady wake time most days
- Reduce screen brightness at night
- Avoid heavy meals too late
- Try a short wind down routine like reading or stretching
This kind of guidance is simple but it works when it is consistent.
Focus learning and cognitive stamina
Focus is not endless. It runs on sleep recovery and mental load. Many people feel they should push harder but the smarter move is often to pace work and plan breaks.
SynapseX could help by:
- Tracking simple attention tasks over time
- Showing how focus changes after poor sleep
- Suggesting a break plan such as 25 minutes work then 5 minutes rest
- Encouraging fewer hard tasks on high strain days
This supports performance without burnout. It also helps students during exam seasons and workers during heavy projects.
Aging well and independence
For older adults small changes can signal a need for extra support. This does not mean panic. It means earlier awareness.
SynapseX could support aging well by:
- Tracking long term trends in sleep activity and simple cognitive tasks
- Providing easy weekly summaries
- Offering optional sharing with family or caregivers with clear consent
- Suggesting check ins when patterns change for weeks
This approach respects independence. It supports early action rather than late crisis.
Stress and mental wellbeing support

Stress affects memory sleep and mood. People often feel stress in the body before they understand it in the mind. Brain health intelligence can help by making patterns visible.
SynapseX could support wellbeing by:
- Allowing simple daily stress check ins
- Connecting stress patterns with sleep and workload
- Offering guided breathing and grounding exercises
- Encouraging professional care when warning signs persist
This is not a replacement for therapy. It is a support tool that can help people act sooner and talk more clearly with professionals.
Evidence trust and ethics
Any tool that touches brain health must be built with strong ethics. People should be able to trust the system. They should also be able to understand it.
SynapseX should show E E A T qualities in clear ways.
Experience means it is built with real user feedback and tested in real settings.
Expertise means clinicians neuroscientists and engineers help shape the product.
Authoritativeness means results are benchmarked and shared with honesty.
Trustworthiness means privacy security and transparency are strong.
Validation and benchmarking
Validation answers a basic question. Does the system work as claimed.
Examples of validation include:
- Testing sleep estimates against strong references
- Testing cognitive tasks for repeatability
- Measuring whether suggestions improve sleep consistency
- Running pilots with clinics to see if the summaries help care conversations
A serious system reports both strengths and limits. That honesty improves trust.
Fairness bias and inclusivity
A model trained on one group may not work well for others. That can lead to unfair outcomes. For example voice based signals can vary by language accent and age.
Fairness practices include:
- Training with diverse datasets
- Measuring performance by subgroup
- Monitoring drift over time
- Giving users ways to report errors and get support
Fairness is not a one time task. It is ongoing.
Transparency people can understand
Users should not need to guess what a score means. SynapseX should explain:
- What each metric measures
- What can influence it
- What is normal variation
- What steps are suggested and why
- When to seek professional help
Good transparency reduces anxiety and improves action.
How organizations can adopt SynapseX
Brain health intelligence is useful for individuals. It can also support clinics research teams and workplace wellness programs when used carefully.
Organizations should adopt it in a way that supports people rather than policing them. Consent must be real and pressure must be avoided.

Pilot programs
A pilot is the safest way to start. A good pilot includes:
- A clear goal such as better sleep consistency in eight weeks
- A small volunteer group
- Simple onboarding and training
- Clear privacy and consent steps
- Regular feedback from participants
The pilot should focus on learning not hype.
Workflow integration
Clinicians and care teams are busy. If the system adds work it will fail. SynapseX should integrate in a light way.
Helpful integration includes:
- Weekly summaries not constant alerts
- Trend highlights with short explanations
- Clear escalation guidance such as when to recommend a visit
- Easy export for reports
Measuring outcomes
Organizations should measure both health outcomes and user experience.
| Area | Example metric | Why it matters |
|---|---|---|
| Sleep | Bedtime consistency trend | Strong link to recovery |
| Stress | Weekly stress check in trend | Shows load changes |
| Cognition | Reaction time trend | Simple stamina signal |
| Engagement | 30 day retention | Proves usefulness |
| Safety | False alert rate | Reduces harm and worry |
| Equity | Participation across groups | Supports fairness |
FAQs
1 What is brain health intelligence used for
It is used to track trends related to sleep stress focus and recovery so people can make better daily choices and seek help sooner when needed
2 Can SynapseX diagnose brain conditions
It should not diagnose conditions unless it is approved and designed as a medical device A responsible system focuses on guidance trends and support
3 What makes a brain tool trustworthy
Clear consent strong privacy protections validation studies transparent explanations and honest limits all build trust
4 How often should I check my results
Most people do well with a short daily check and a weekly review Trends matter more than one day
5 What if my data looks worse for a few days
That can happen due to travel illness stress or poor sleep Look for patterns over weeks If the change persists consider speaking with a professional
6 Is it safe to share my data with family or caregivers
It can be safe if sharing is optional controlled by you and protected by strong security You should be able to stop sharing at any time
7 How can organizations use this without harming trust
They should use volunteers protect privacy avoid punishment based on data and focus on support and wellbeing
Conclusion
Brain health deserves everyday attention not just emergency attention. With responsible design and strong ethics brain health intelligence can help people understand their patterns early and take simple steps that protect focus calm and recovery.
SynapseX represents a future where brain insights are clear supportive and secure. The best outcome is not perfect scores. The best outcome is better days more often and earlier help when it is needed.
Related Articles
Why Mental Health Matters and Simple Ways to Care for It
Healthy Lifestyle for Better Memory and Focus: Complete Brain Health Guide
Science/Health
Train Your Brain for Peak Performance: Habits That Boost Focus and Mental Clarity

Modern life moves fast, and it can feel like your attention gets pulled in ten directions at once. The good news is you can train your brain for peak performance without fancy tools or extreme routines. With a few steady habits, you’ll build focus and mental clarity that lasts through school, work, family time, and everything in between.
This guide is practical and realistic. It uses small steps, because small steps actually stick. You’ll also see that focus isn’t just “willpower.” It’s a mix of energy, environment, and repeatable systems.
What Peak Performance Really Means in Daily Life
Peak performance doesn’t mean working nonstop or acting like a robot. It means you can do the right task at the right time with less stress. It also means you recover well, so you can show up again tomorrow.
When you train your brain for peak performance, you’re aiming for:
- Better attention span
- Faster task switching when needed (without getting stuck)
- Clear thinking under pressure
- Steadier mood and patience
- More consistent results, even on “meh” days
The brain basics: attention, working memory, and energy
To keep things simple, focus depends on three big pieces:
- Attention: what you choose to notice and stay with
- Working memory: your brain’s scratchpad that holds info briefly
- Energy: mental fuel, affected by sleep, food, stress, and movement
If one piece drops, your performance drops too. That’s why habits work so well: they support all three at once.
Why habits beat motivation most days
Motivation comes and goes. Habits stay. When you set up a routine, you don’t have to debate with yourself every time. You just do the next step. Over time, you train your brain for peak performance by making the helpful choice the easy choice.

Habit 1: Build a Focus-Friendly Morning Start
A strong morning doesn’t need to be long. It needs to be consistent. If your first hour is chaotic, your brain often stays in “reactive mode” all day.
Use a simple first 15 minutes routine
Here’s a routine that’s short but powerful:
- Stand up and open a window or get light on your face
- Drink water
- Do one small stretch (30 seconds is fine)
- Write your top 3 priorities for the day
That’s it. No drama. No perfection.
Light, water, and a quick plan
These steps matter because:
- Light helps signal wake up to your brain
- Water supports alertness and reduces sluggishness
- A quick plan reduces mental clutter and improves mental clarity
If you want to train your brain for peak performance, start by training your morning to be calmer than your notifications.
A short checklist you can reuse
Use this checklist for two weeks:
| Morning item | Time needed |
| Water | 1 minute |
| Light exposure | 2–5 minutes |
| Stretch or walk | 1–5 minutes |
| Write top 3 tasks | 3 minutes |
| Put phone on silent | 10 seconds |
Habit 2: Work in Sprints Then Recover on Purpose
Many people try to focus by forcing long hours. That usually backfires. Your brain works better in cycles.

The 25–5 and 50–10 methods
Two simple sprint options:
- 25–5: work 25 minutes, break 5 minutes
- 50–10: work 50 minutes, break 10 minutes
During the sprint, you do one task only. During the break you recover on purpose. This is one of the simplest ways to train your brain for peak performance because it teaches sustained attention without burnout.
Micro-break ideas that actually reset your mind
Good breaks are not scrolling breaks. Try:
- Stand and breathe slowly for 60 seconds
- Look far away out a window (rest your eyes)
- Refill water
- Walk to another room and back
- Do 10 slow shoulder rolls
These micro-breaks lower stress and bring back mental clarity.
A sample sprint schedule (table)
| Time block | What you do | Why it helps |
| 9:00–9:25 | Deep work sprint | Builds focus |
| 9:25–9:30 | Walk + water | Restores energy |
| 9:30–9:55 | Deep work sprint | Keeps momentum |
| 9:55–10:05 | Longer break | Prevents fatigue |
| 10:05–10:55 | Longer task sprint | Better output |
Habit 3: Eat and Hydrate for Steady Brain Power
Food affects attention more than most people think. Big sugar spikes can lead to crashes. Skipping meals can make you foggy and irritable. If you want to train your brain for peak performance, steady fuel matters.
Balanced meals that support attention
Aim for meals with:
- Protein (eggs, tofu, fish, chicken, beans)
- Fiber (vegetables, fruit, oats, brown rice)
- Healthy fats (nuts, olive oil, avocado)
This mix supports stable energy which supports focus and mental clarity.
Quick meal ideas:
- Oats + yogurt + fruit
- Rice + veg + tofu or chicken
- Wholegrain sandwich + egg + salad
- Soup + beans + side of fruit
Hydration cues and a practical target
Instead of complex rules use cues:
- Drink water when you wake up
- Drink water before your first big task
- Drink water at lunch
- Drink water mid-afternoon
A practical target is enough to keep your urine light yellow most of the day. If it’s dark, you likely need more water.
Smart caffeine timing (and when to stop)
Caffeine can help focus but timing matters:
- Try waiting 60–90 minutes after waking before caffeine
- Avoid caffeine 8 hours before bedtime if sleep is affected
This keeps your sleep stronger and sleep is a major driver when you train your brain for peak performance.
Habit 4: Train Your Attention Like a Skill

Attention is trainable. You don’t need to be born focused. You can practice it the same way you practice a sport or a musical instrument.
One-tasking rules that make focus easier
Try these one-tasking rules:
- Keep only one tab or document open for the task
- Put your phone out of reach
- If you think of another task write it on a note and return
It sounds basic, but it’s powerful. Multitasking is usually just fast distraction.
A 2-minute reset for mental clarity
Use this reset when you feel scattered:
- Sit up straight
- Inhale slowly for 4 seconds
- Exhale slowly for 6 seconds
- Repeat for 2 minutes
- Then write one sentence: My next best step right now is ____.
This reset helps your nervous system calm down, which improves mental clarity quickly.
Simple mindfulness that doesn’t feel fluffy
Mindfulness can be practical:
- While washing your hands, feel the water and count to 10
- While walking, notice 5 things you can see
- While eating, take three slow bites before doing anything else
These moments train attention gently. Over time, you train your brain for peak performance by teaching it to stay present on purpose.
Habit 5: Sleep as Your Secret Performance Tool
Sleep is not a luxury. It’s part of your brain’s maintenance system. If you skip it, your focus drops, your mood gets touchy, and your mental clarity fades.
A consistent sleep window
Aim to go to bed and wake up at about the same time every day, including weekends. Your brain loves rhythm. A stable rhythm helps you train your brain for peak performance because your energy becomes more predictable.
A simple goal:
- Same bedtime within 60 minutes each night
- Same wake time within 60 minutes each morning
Screens light, and wind-down habits
To make sleep easier:
- Dim lights 60 minutes before bed
- Keep your phone out of reach at night, or switch to a basic alarm clock instead.
- Choose calm activities: reading, light stretching, journaling
If you can’t fall asleep, don’t panic. Get up, do something boring in low light, then try again.
A quick sleep-friendly checklist
- Stop heavy meals close to bedtime
- Keep the room cool and dark
- Write tomorrow’s top 3 tasks before bed
- If worried, write a worry list and park it
That last step is surprisingly helpful for mental clarity.
Habit 6: Design Your Environment to Remove Friction
Your environment can either support focus or sabotage it. If distractions are always within reach, you’ll use willpower all day. That’s exhausting.
Desk setup noise and phone placement
Try these environment upgrades:
- Put your phone behind you or in a drawer
- Keep the desk surface simple: only today’s materials
- Use noise control if needed (quiet room, earplugs or steady background sound)
These changes reduce decision fatigue and help you train your brain for peak performance without extra effort.
If-then plans for distractions
Make tiny rules ahead of time:
- If I want to check social media then I write it down and wait for my next break
- If I feel stuck then I work for 5 more minutes before changing tasks
- If someone interrupts then I say Give me 10 minutes and I’ll come back.
These plans sound simple, but they protect your attention.
A workspace audit list
Check your workspace once a week:
- Are cords, papers, and clutter creeping back?
- Is your chair comfortable enough?
- Are your most-used items within reach?
- Are distracting apps pinned on your screen?
Small fixes keep your system strong.
For a deeper look at daily habits that support attention, read my post Healthy Lifestyle for Better Memory and Focus: Complete Brain Health Guide.

FAQs
1) How long does it take to improve focus and mental clarity?
Many people notice small changes within a week especially from better sleep and fewer distractions. Bigger changes often take 4–8 weeks because habits need repetition to stick.
2) What’s the fastest way to train your brain for peak performance?
Start with two actions: a consistent sleep schedule and sprint-based work sessions. These improve energy and attention quickly and make other habits easier.
3) Is music good or bad for focus?
It depends on the task. For reading or writing, lyrics can distract. For repetitive work, steady background sound may help. If your output drops, switch to silence.
4) What if my mind keeps wandering during work sprints?
That’s normal. Use a note page called Later. Each time a thought pops up, write it down and return to the task. This keeps mental clarity without fighting your brain.
5) How do I avoid afternoon brain fog?
Eat a balanced lunch, drink water, and take a short walk or stretch break. Also, avoid heavy sugary snacks that can cause an energy crash.
6) Can stress reduce my ability to focus?
Yes. Stress raises mental noise and makes it harder to think clearly. Breathing resets, short walks, and planning your next step can lower stress and restore focus.
7) Should I do brain games to improve attention?
Brain games can be fun, but daily habits matter more. Sleep, movement, nutrition, and distraction control usually give bigger results in real life.
Conclusion: Your Next 7 Days Plan
If you want results, keep it simple. For the next 7 days, do this:
- Start your day with water, light, and a top-3 list
- Do two work sprints daily with real breaks
- Keep your phone out of reach during sprints
- Eat one balanced meal each day and drink water at set times
- Keep a consistent sleep window
These steps are realistic, and they add up fast. When you train your brain for peak performance, you’re not chasing perfection. You’re building a steady system that makes focus and mental clarity more natural every week.
Science/Health
Healthy Lifestyle for Better Memory and Focus: Complete Brain Health Guide

If you’ve ever walked into a room and forgotten why, or stared at your screen unable to focus, you’re not alone. In today’s fast-paced world, a healthy lifestyle for better memory and focus isn’t just a bonus it’s a basic need.
The good news is your brain isn’t fixed. It changes with your habits, your environment, and even your thoughts. This guide explains, in simple language, how everyday choices around food, sleep, movement, stress, and technology can sharpen your mind and protect your brain for life.
Understanding Brain Health, Memory, and Focus
How Your Brain Works Day to Day
Your brain is like the control centre of a busy city. Billions of cells called neurons send messages using tiny electrical and chemical signals. These messages help you:
- Pay attention in conversations
- Remember names, passwords, and directions
- Plan your work or studies
- Control mood and motivation
Two key processes matter for memory and focus:
- Neuroplasticity – your brain’s ability to change and form new connections
- Neurogenesis – creation of new brain cells, especially in the memory area (hippocampus)
A healthy lifestyle for better memory and focus strengthens both of these processes, so your brain becomes more adaptable and resilient.
Types of Memory You Use All the Time
You use different types of memory without even noticing:
- Short-term (working) memory – holding a phone number long enough to dial it
- Long-term memory – facts, skills, and life events
- Procedural memory – how to ride a bike or type on a keyboard
Focus is like the gatekeeper. When you focus well, more information passes from short-term to long-term memory. When you’re distracted or exhausted, that gate doesn’t work properly, and things just don’t stick.
To learn more about emotional wellbeing and everyday coping tools, check out our guide on Why Mental Health Matters and Simple Ways to Care for It.
Why a Healthy Lifestyle Matters for Better Memory and Focus

The Brain Body Connection
Your brain isn’t floating on its own. It depends on:
- Blood flow to bring oxygen and nutrients
- Hormones to balance mood and energy
- Immune system to control inflammation
When you choose a healthy lifestyle for better memory and focus, you’re really improving:
- Heart and blood vessel health
- Blood sugar control
- Inflammatory levels in the body
All of these directly affect your ability to think clearly and remember.
Common Threats to Brain Health
Many everyday habits quietly harm your brain over time:
- Chronic sleep loss – breaks down attention and memory consolidation
- High-sugar, ultra-processed diet – linked to inflammation and slower thinking
- Long-term stress damages brain areas involved in memory
- Smoking and heavy alcohol use – increase risk of stroke and dementia
- Lack of movement – reduces blood flow and brain growth factors
Some risks are especially important in urban settings like Singapore, where busy jobs, exam pressure, and screen-heavy lifestyles are common.
Nutrition for a Sharper Brain
A key part of a healthy lifestyle for better memory and focus is what’s on your plate. Food is raw material for brain cells and the fuel that powers your thoughts.
Key Brain-Boosting Nutrients
Your brain uses about 20% of your daily energy. It needs:
- Omega‑3 fatty acids (EPA, DHA) – support cell membranes and signalling
- Antioxidants (vitamins C, E, polyphenols) – protect from damage
- B vitamins (B6, B9, B12) – help with energy and neurotransmitters
- Magnesium and zinc – support learning and memory
- Choline – needed for acetylcholine, a memory chemical
A balanced diet usually covers these, but many people fall short, especially on omega‑3s and fibre.
Best Foods for Memory and Focus
Think in terms of patterns, not single “superfoods.” Aim for:
- Fatty fish – salmon, sardines, mackerel (rich in omega‑3)
- Leafy greens – spinach, kailan, bok choy, kangkong
- Colourful fruits and vegetables – berries, oranges, papaya, capsicum
- Whole grains – brown rice, wholemeal bread, oats, quinoa
- Nuts and seeds – walnuts, almonds, chia, flaxseed
- Legumes – lentils, chickpeas, soy beans, tau kwa
- Healthy fats – olive oil, avocado, nuts
Try to limit:
- Sugary drinks and desserts
- Deep-fried fast foods
- Processed meats and instant noodles
These can raise inflammation and blood sugar spikes, which hurt focus.
Simple Meal Ideas for Busy People
If you’re busy with work or school, you can still support brain health:
Breakfast
- Oatmeal with berries, a spoon of ground flaxseed, and a boiled egg
- Wholemeal toast with avocado and sliced tomato
Lunch
- Brown rice with grilled fish, mixed vegetables, and tofu
- Salad bowl with leafy greens, chickpeas, nuts, and olive oil dressing
Dinner
- Stir-fried vegetables with tofu and a small portion of noodles or rice
- Baked salmon with sweet potato and broccoli
Smart snacks
- A small handful of nuts
- Fruit with yoghurt
- Carrot sticks with hummus
If you eat out often (very common in Singapore), you can still choose:
- More vegetables and clear soups
- Less gravy, less deep-fried items
- Water or unsweetened tea instead of sugary drinks

Hydration, Caffeine, and Smart Beverage Choices
Why Water Intake Affects Thinking Speed
Even mild dehydration – just 1–2% of your body weight – can:
- Reduce attention
- Slow reaction time
- Increase fatigue and headaches
For better memory and focus, keep a water bottle near you. Plain water is best, but unsweetened herbal or green tea also helps.
Signs you may not be drinking enough:
- Dark yellow urine
- Dry mouth
- Frequent headaches
- Feeling sluggish for no clear reason
In a warm, humid climate like Singapore’s, you may need more fluids, especially if you’re walking outside or exercising.
Using Caffeine Without the Crash
Caffeine can help:
- Improve alertness
- Boost reaction time
- Increase motivation for tasks
But too much:
- Worsens anxiety
- Disrupts sleep (which harms memory)
- Can cause energy crashes
Use caffeine wisely:
- Limit to about 1–2 cups of coffee or tea per day
- Avoid strong caffeine after mid-afternoon
- Skip sugary energy drinks – the sugar spike hurts focus later
If you’re sensitive, try green tea: it has less caffeine and L‑theanine, which may support calm focus.
Sleep: The Night-Time Secret of Strong Memory
What Happens in Your Brain While You Sleep
During good-quality sleep, your brain:
- Sorts and stores new memories
- Clears waste products linked to brain diseases
- Resets emotional centres
- Repairs cells and connections
If you cut sleep short, you’re not just tired. You’re blocking a core part of a healthy lifestyle for better memory and focus.
Chronic sleep loss leads to:
- Poor attention and slower thinking
- Weaker learning and memory
- Higher risk of mood problems and burnout
Building a Sleep Routine for Better Focus
Aim for 7–9 hours of sleep most nights. To improve sleep:
- Keep a regular schedule – same sleep and wake times, even on weekends
- Wind down routine – 30–60 minutes with low light and calm activities (reading, stretching)
- Limit screens before bed – blue light can trick your brain into staying awake
- Avoid heavy meals and alcohol close to bedtime
- Keep your room cool, dark, and quiet
If racing thoughts keep you up, try:
- Writing a quick “worry list” and parking it for tomorrow
- Simple breathing: in for 4 seconds, hold 4, out for 6–8 seconds
If snoring, gasping, or very unrefreshing sleep is common, consider seeing a doctor. Sleep disorders like sleep apnea are treatable and strongly affect brain health.
Exercise and Movement for Long-Term Brain Health

How Physical Activity Changes the Brain
Exercise isn’t just for muscles. It’s one of the strongest tools in a healthy lifestyle for better memory and focus. Regular movement:
- Increases blood flow and oxygen to the brain
- Triggers release of BDNF (brain-derived neurotrophic factor), a “fertiliser” for brain cells
- Supports new connections and helps protect against age-related decline
- Improves mood and reduces stress, which helps focus
Practical Workout Ideas for Every Fitness Level
You don’t need a gym membership to support your brain. Try to get:
- 150 minutes per week of moderate exercise (like brisk walking)
- Plus strength training 2 days per week
Simple ways to do this:
- Walk 30 minutes most days (or 3 × 10-minute walks)
- Take stairs when possible
- Do bodyweight exercises at home: squats, push-ups against the wall, planks
For more variety:
- Join a dance, martial arts, or yoga class
- Cycle in the park
- Swim a few laps at the pool
If you’re starting from zero, begin small:
- 5–10 minutes of walking daily
- Gradually add time and intensity
Any increase in movement is better than none. Consistency matters more than perfection.
Stress, Emotions, and Mental Clarity
How Stress Blocks Memory and Focus
Short bursts of stress can sharpen you. But chronic stress:
- Raises cortisol levels
- Shrinks brain areas linked to memory (like the hippocampus)
- Makes you more easily distracted and forgetful
You might notice:
- Trouble concentrating on reading or meetings
- Forgetting simple tasks
- Feeling mentally “foggy”
A healthy lifestyle for better memory and focus must include stress management, not just food and exercise.
Simple Daily Stress-Management Tools
You don’t need fancy equipment to calm your mind:
- Deep breathing – a few slow breaths can lower stress signals
- Mindfulness – paying attention to the present moment without judging it
- Journaling – writing down worries or gratitude
- Nature time – short walks in green spaces can improve mood and clarity
- Social support – talking with friends or family members you trust
Try this simple practice:
- Sit comfortably, close your eyes
- Inhale slowly for 4 seconds
- Exhale gently for 6–8 seconds
- Repeat for 1–3 minutes
Over time, you’ll train your nervous system to relax more easily.
If stress feels overwhelming or lasts for weeks, consider professional help. Counsellors and psychologists can teach coping skills and spot problems like anxiety or depression.
Digital Habits and Focus in a Hyper-Connected World
The Cost of Constant Notifications
Smartphones and laptops are powerful tools, but constant alerts can:
- Break your attention every few minutes
- Make tasks take longer
- Increase stress and mental fatigue
Your brain needs “deep work” time – blocks of focused attention to learn and remember well. Without it, even a strong healthy lifestyle for better memory and focus can’t work at full power.
Creating a Brain-Friendly Digital Environment

Simple changes can boost focus:
- Turn off non-essential notifications
- Turn on Do Not Disturb when you need to focus on important work or study.
- Check email or messages at set times instead of all day
- Keep your phone out of sight when concentrating
Try the Pomodoro technique:
- Work for 25 minutes with full focus
- Take a 5-minute break
- After 4 rounds, take a longer 15–30-minute break
During breaks, avoid scrolling social media. Instead:
- Stand up and stretch
- Take a few deep breaths
- Get a drink of water
Your brain will thank you with better concentration and memory.
Brain-Training, Learning, and Mental Fitness
Does Brain Training Really Work?
Apps and games that claim to boost memory and focus can help in some ways, but they’re not magic. They often improve your skill at the game itself more than overall thinking.
For a truly healthy lifestyle for better memory and focus, mix in real-world mental challenges:
- Learning a new language
- Playing a musical instrument
- Tackling difficult books or courses
- Solving real problems at work or in your community
These activities build broad skills like planning, attention, and flexible thinking.
Lifelong Learning Strategies for a Strong Mind
To keep your brain fit as you age:
- Stay curious – ask questions, read widely
- Teach others – explaining a topic strengthens your own understanding
- Space your learning – study a bit each day rather than cramming
- Mix topics – combine logic tasks with creative ones
You don’t have to study something huge. Even small, steady learning like reading 10 pages a day or watching an educational video – adds up over years.
Everyday Habits That Protect Your Brain Long-Term
Safety, Medical Conditions, and Check-Ups
Brain health also depends on physical safety and medical care:
- Protect your head – use helmets for cycling, motorbiking, or some sports
- Manage blood pressure, diabetes, and cholesterol these affect blood flow to your brain
- Don’t smoke, it damages blood vessels and raises stroke risk
- Keep alcohol intake low, as heavy drinking can damage memory and clear thinking
Routine visits to your doctor can detect potential problems at an early stage. If you notice sudden changes in memory or focus, don’t ignore them.
Social Life, Hobbies, and Purpose
Humans are social, Strong relationships and meaningful activities are part of a healthy lifestyle for better memory and focus:

- Spend time with friends and family
- Join clubs, interest groups, or volunteer programmes
- Keep hobbies alive, gardening, cooking, reading, singing, anything you enjoy
Having a sense of purpose, something you care about and work towards can:
- Protect against depression
- Support healthy habits
- Give your brain reasons to stay active and engaged
FAQs on Healthy Lifestyle for Better Memory and Focus
1. How long does it take to see results from a healthy lifestyle for better memory and focus?
Some people notice better focus within 1–2 weeks, especially after improving sleep and cutting down on junk food. Bigger changes in memory and mood may take 1–3 months of steady habits.
2. Can a healthy lifestyle for better memory and focus prevent dementia?
No lifestyle can guarantee prevention, but strong habits can lower your risk and may delay symptoms. Exercise, good nutrition, social contact, and mental activity all help protect the brain.
3. Do I need supplements for better memory and focus?
Most people can get what they need from a balanced diet.Supplements can be useful if you have a deficiency (such as vitamin B12 or vitamin D), but they should never be seen as a substitute for a complete healthy lifestyle for better memory and focus. Consult a healthcare professional before beginning any supplement.
4. Is coffee good or bad for brain health?
In moderate amounts, coffee or tea can support alertness and focus. Too much can disturb sleep and increase anxiety, which harms memory. Try to keep caffeine moderate and avoid it late in the day.
5. I’m very busy. What’s the single best change I can start with?
If you’re overloaded, start with sleep. Aim for 7–8 hours per night, with a regular bedtime and wake time. Consistent sleep powers your brain, and once you feel better, it becomes easier to tackle food, exercise, and other habits.
6. How can I help my child or teenager improve memory and focus?
For young people, the same principles apply:
- Regular sleep schedule
- Nutritious meals and snacks
- Time outdoors and daily movement
- Limit on screen time, especially near bedtime
- Plan your study into shorter sessions with regular breaks instead of long cramming marathons
Model the habits yourself. Children are more likely to imitate what you do than follow what you say.
Conclusion: Putting Your Brain-Healthy Lifestyle Into Action
Your brain is your most valuable asset. A healthy lifestyle for better memory and focus isn’t about perfection; it’s about many small choices repeated day after day.
To get started, pick just one or two of these steps:
- Include one more portion of vegetables with your main meal.
- Walk 15–20 minutes daily
- Turn off non-essential phone notifications
- Stick to a consistent bedtime every night
- Practice 3 minutes of deep breathing during your day
As these become habits, add more. Over months and years, these small changes build a powerful shield for your brain, supporting clearer thinking, stronger memory, and better focus at every age.
Read More on Sgtrends
-
Blog9 months ago
Wedding day horror: Groom falls to death at VOCO Orchard Hotel on Wedding Day
-

 Technology8 months ago
Technology8 months agoEdTech in Singapore: 150+ Companies Driving $180M Digital Learning Revolution
-

 Education10 months ago
Education10 months agoSingapore JC Ranking: A Complete Guide for Students
-

 Food7 months ago
Food7 months agoYi Dian Dian Bubble Tea Singapore: Menu, Outlets & Try Drinks
-

 Blog11 months ago
Blog11 months agoBest of SG with Singapore Rediscover Voucher
-

 Business11 months ago
Business11 months agoWhat to Expect During PSLE Marking Days 2023
-

 Digital Marketing7 months ago
Digital Marketing7 months agoHow to Run Effective Xiaohongshu Advertising Campaigns
-

 Digital Marketing7 months ago
Digital Marketing7 months agoWhy Xiaohongshu Ads Are a Must for Reaching Chinese Consumers





